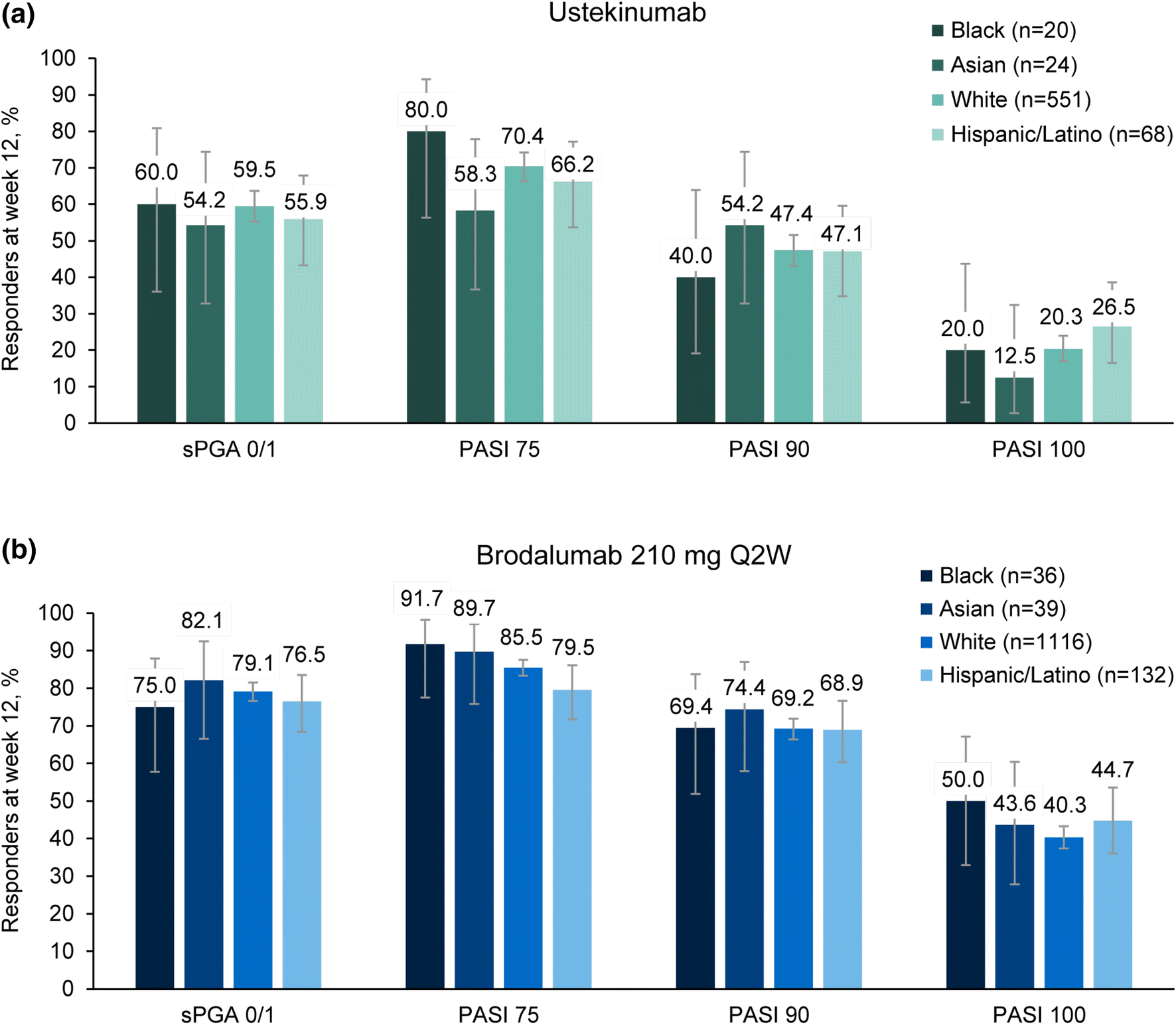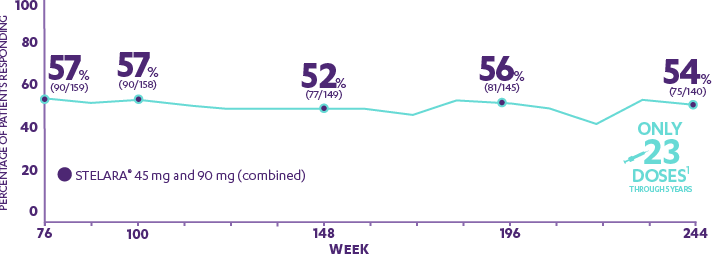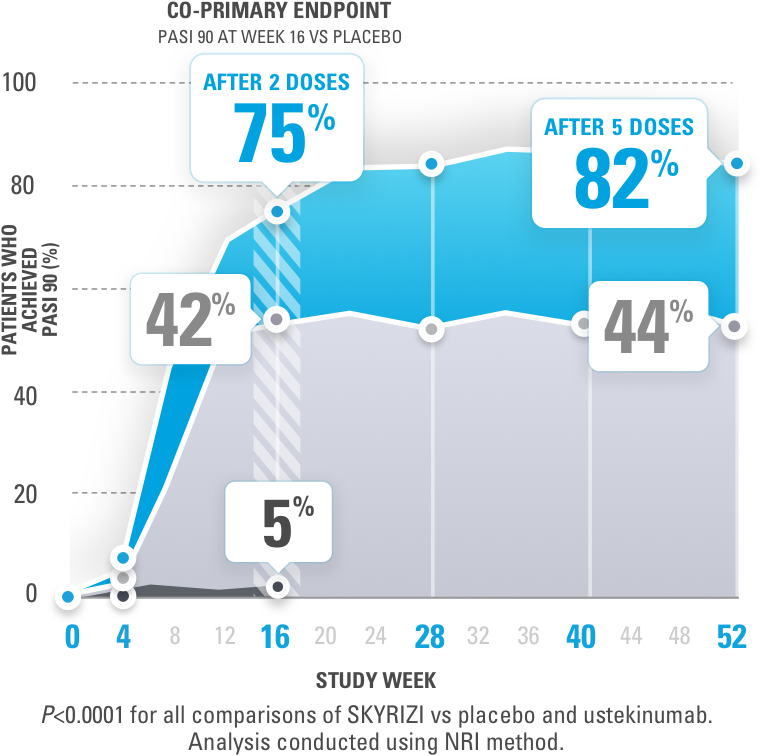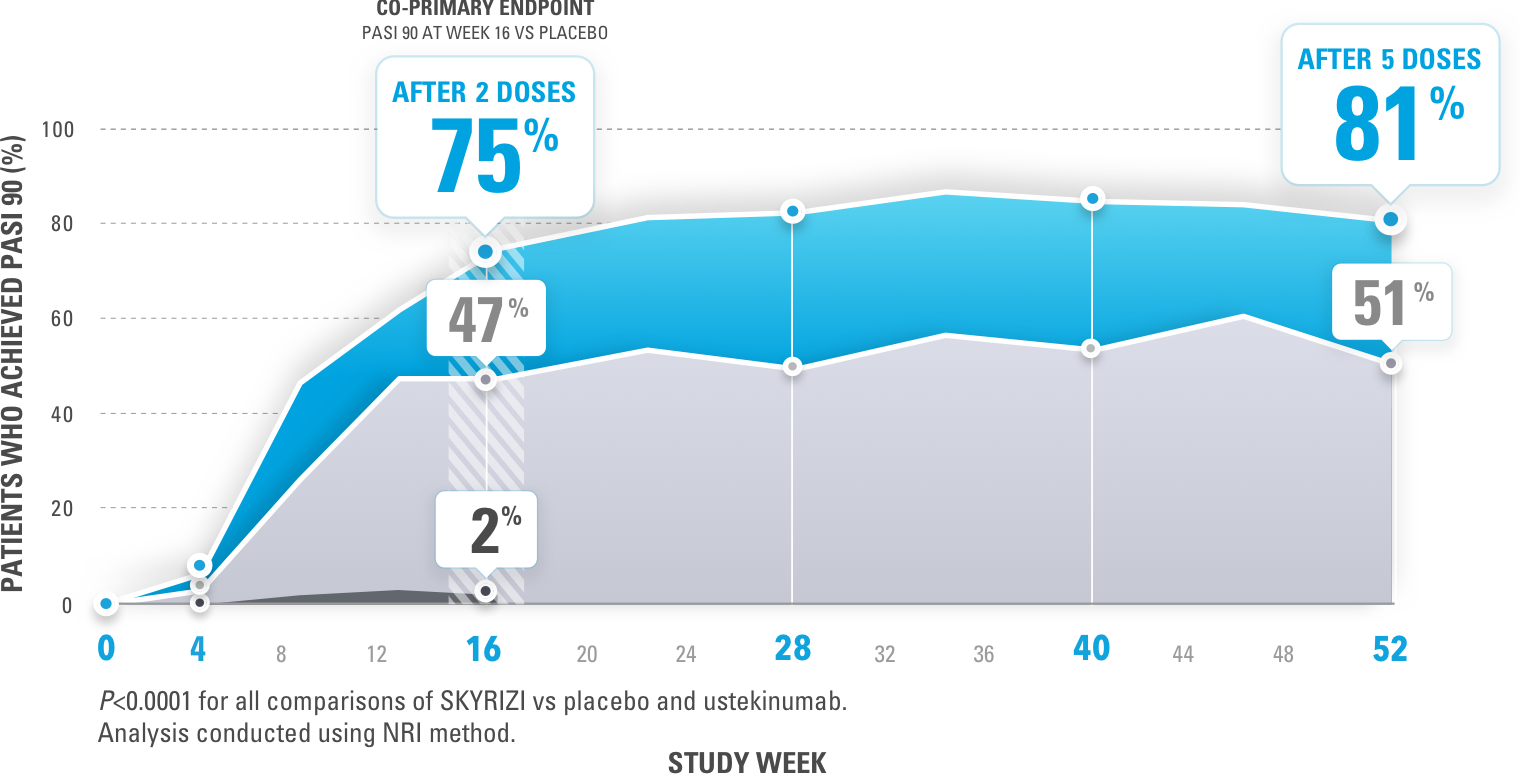
Early Treatment Targets for Predicting Long-term Dermatology Life Quality Index Response in Patients with Moderate-to-Severe Plaque Psoriasis: A Post-hoc Analysis from a Long-term Clinical Study – JCAD | The Journal of Clinical

PASI 75, PASI 90, and PASI 100 response by baseline psoriasis severity... | Download Scientific Diagram

Guselkumab Was More Effective Than Secukinumab in Patients with Plaque Psoriasis and the Subset of Patients with Self-Reported Psoriatic Arthritis in a Randomized, Double-blind, Head-to-head Comparison Study over 1 Year - ACR

Estimated relationships for PaSI90, PaSI100, and sPGa0/1 responses at... | Download Scientific Diagram
Real-world efficacy of biological agents in moderate-to-severe plaque psoriasis: An analysis of 75 patients in Taiwan | PLOS ONE

Percentage of PASI 75, PASI 90, PASI 100 and PGA responders at weeks 12... | Download Scientific Diagram

Efficacy and safety of secukinumab in Chinese patients with moderate-to-severe plaque psoriasis: a real-life cohort study | Chinese Medical Journal

A 50% reduction in the Psoriasis Area and Severity Index (PASI 50) is a clinically significant endpoint in the assessment of psoriasis - ScienceDirect
PASI ≤ 2 corresponds to PASI 90, irrespective of baseline severity: A post-hoc analysis of the PRIME-study

Absolute Versus Relative Psoriasis Area and Severity Index in Clinical Practice | Actas Dermo-Sifiliográficas

Figure 3 | Exposure–Response Relationships for the Efficacy and Safety of Risankizumab in Japanese Subjects with Psoriasis | SpringerLink