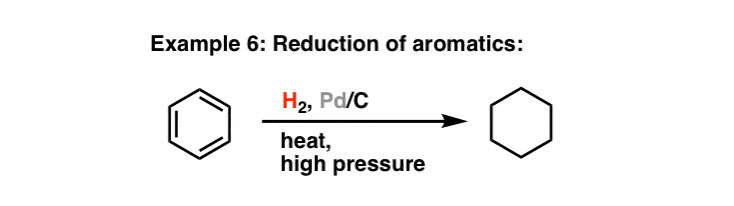
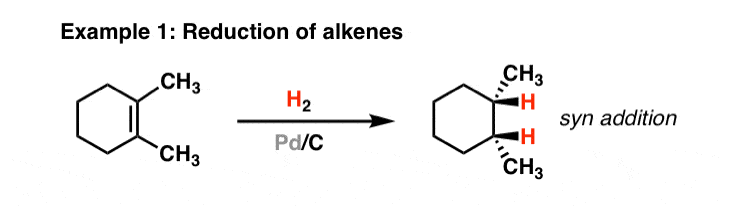
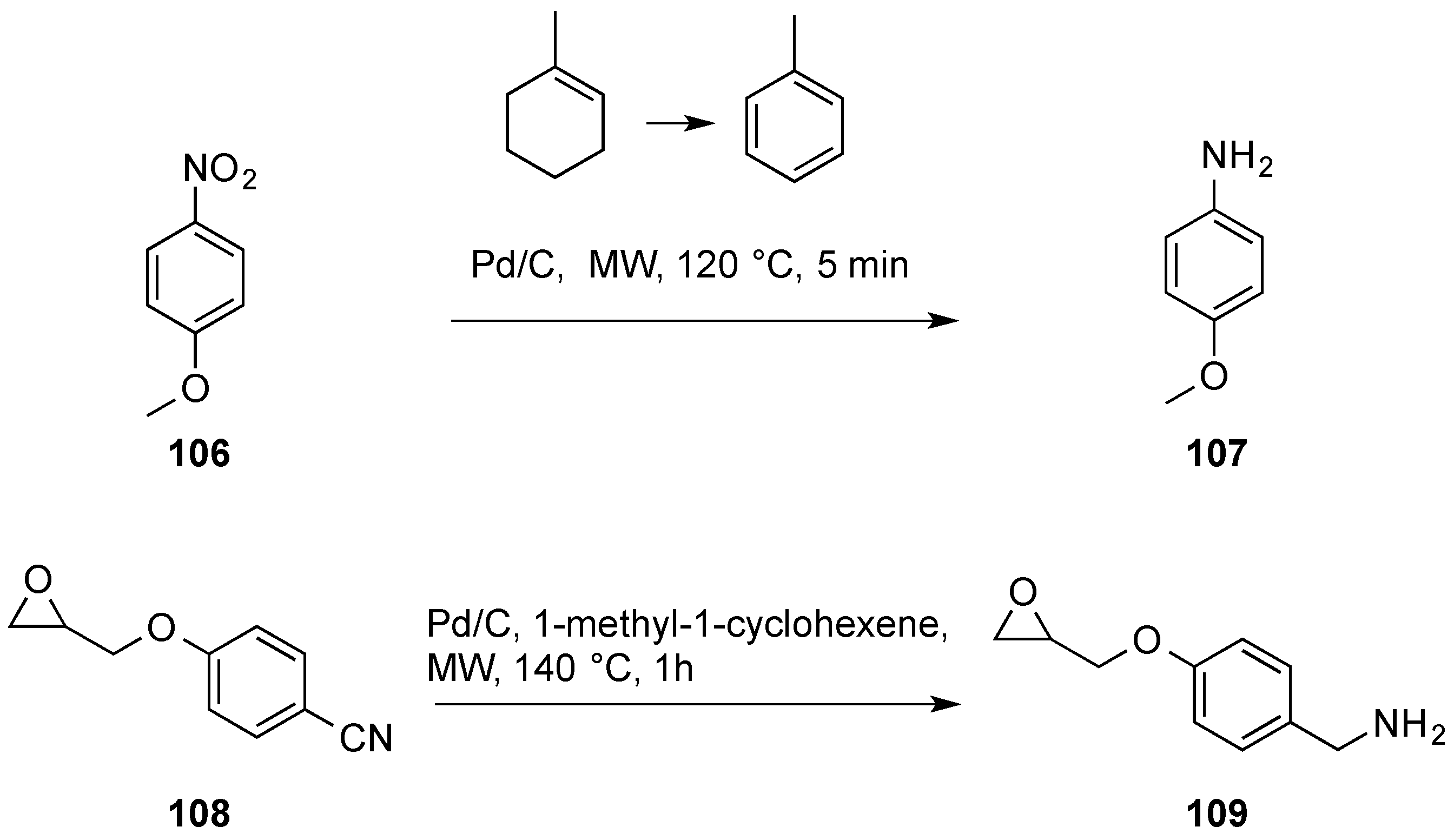
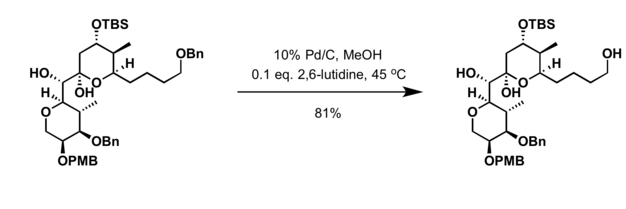
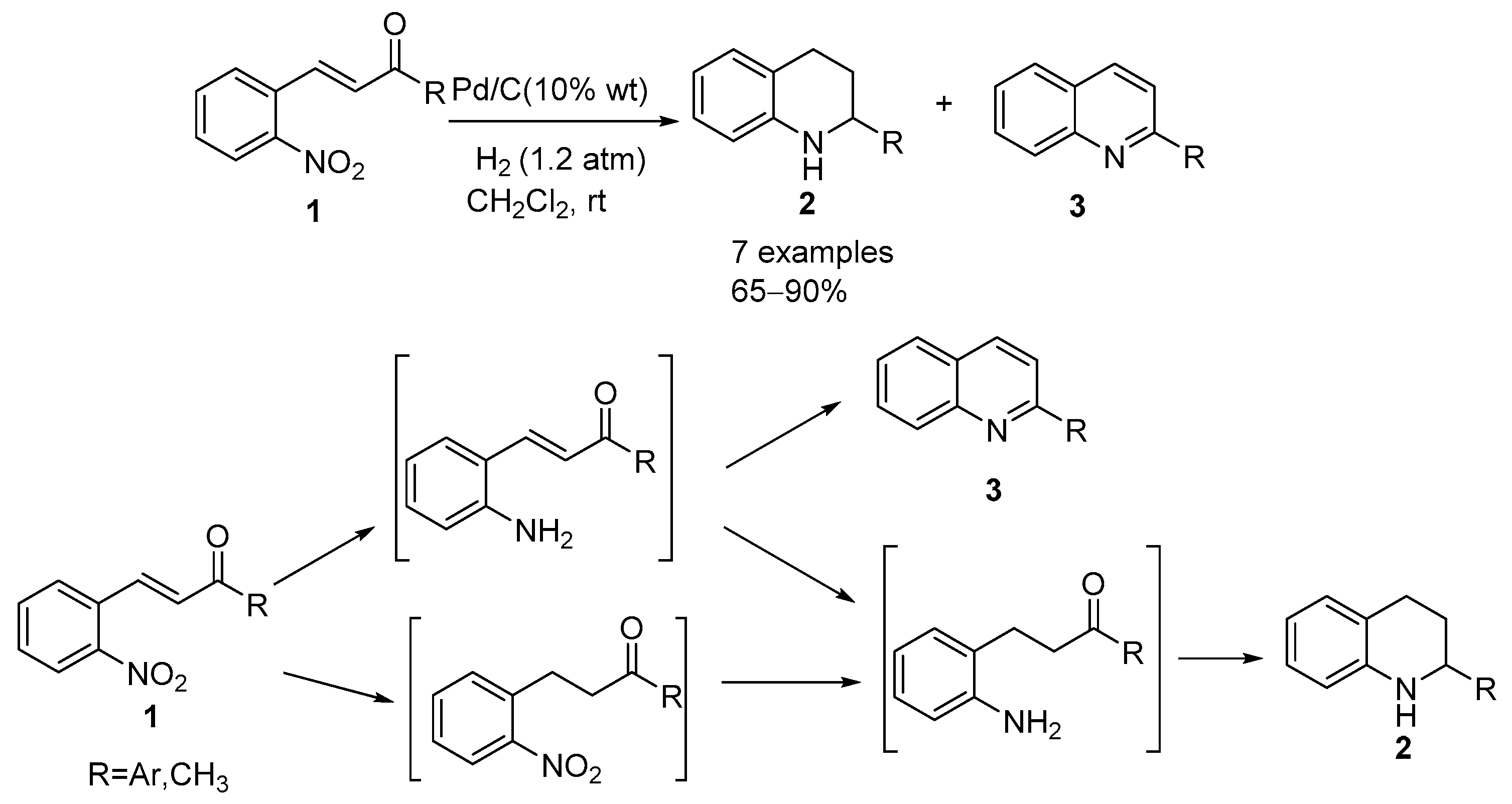
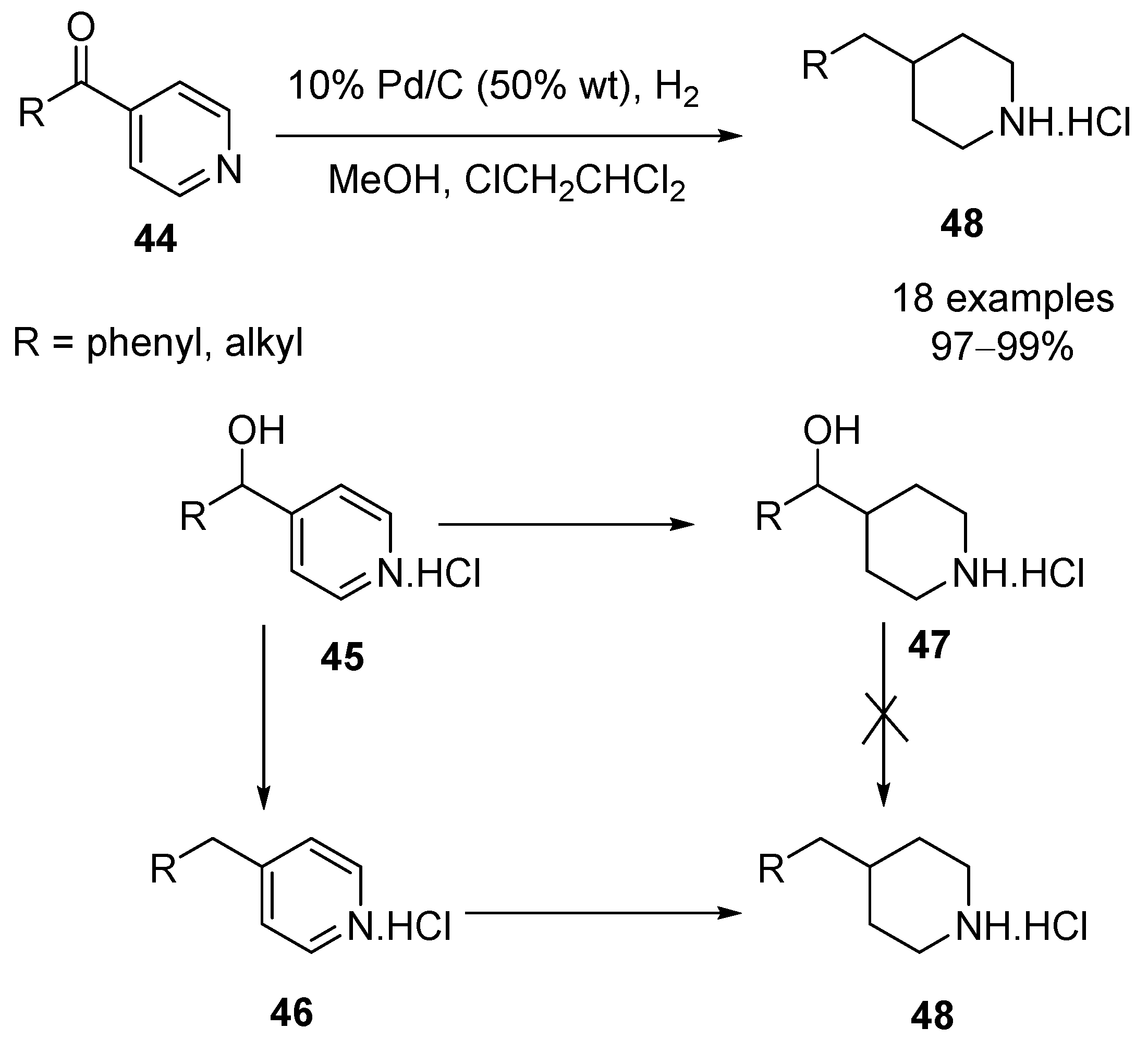
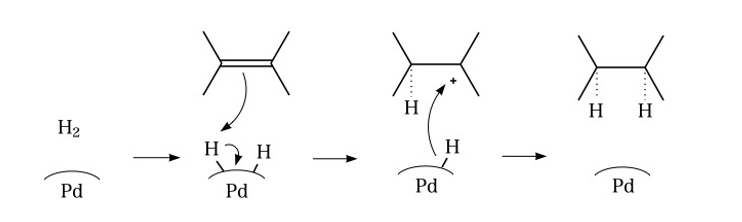
Chemoselective control of hydrogenation among aromatic carbonyl and benzyl alcohol derivatives using Pd/C(en) catalyst - ScienceDirect

Pd/C and NaBH4 in Basic Aqueous Alcohol: An Efficient System for an Environmentally Benign Oxidation of Alcohols

Hydrogenolysis. Reagents and conditions: (a) H2, Pd/C, EtOAc, rt, 24 h,... | Download Scientific Diagram

Influence of Solvents and Additives on the Pyrophoricity of Palladium on Carbon Catalyst after Hydrogenation | Organic Process Research & Development
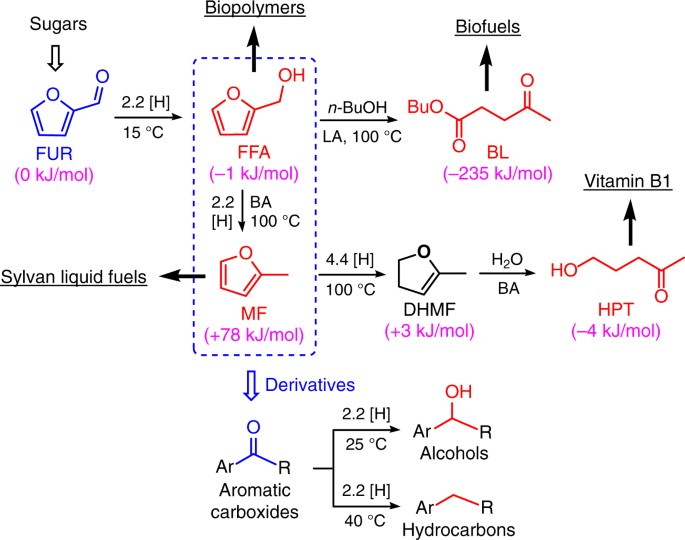
Control of selectivity in hydrosilane-promoted heterogeneous palladium-catalysed reduction of furfural and aromatic carboxides | Communications Chemistry
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img03.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

Hydrogenolysis. Reagents and conditions: (a) H2, Pd/C, EtOAc, rt, 24 h,... | Download Scientific Diagram

Kaili101. Palladium carbon (Pd/C)Carrier catalysts_Carrier catalysts_Kaili Catalyst New Materials CO., LTD
![Pd/C, PEタイプ (Pd 10%)(含水) 【エヌ・イー ケムキャット社製造】・Pd/C, type PE (Pd 10%)(wetted with water) [Manufacturer:N.E. CHEMCAT] ・163-27041・161-27042・169-27043【詳細情報】|【合成・材料】|試薬-富士フイルム和光純薬 Pd/C, PEタイプ (Pd 10%)(含水) 【エヌ・イー ケムキャット社製造】・Pd/C, type PE (Pd 10%)(wetted with water) [Manufacturer:N.E. CHEMCAT] ・163-27041・161-27042・169-27043【詳細情報】|【合成・材料】|試薬-富士フイルム和光純薬](https://labchem-wako.fujifilm.com/sc/05/163-27041.png)
Pd/C, PEタイプ (Pd 10%)(含水) 【エヌ・イー ケムキャット社製造】・Pd/C, type PE (Pd 10%)(wetted with water) [Manufacturer:N.E. CHEMCAT] ・163-27041・161-27042・169-27043【詳細情報】|【合成・材料】|試薬-富士フイルム和光純薬
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img02.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
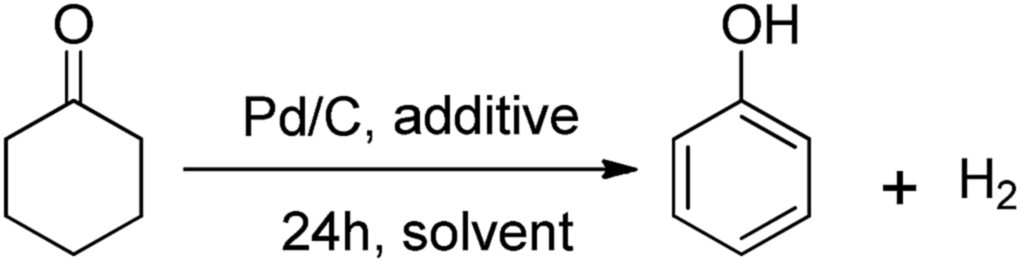
Reaction-activated palladium catalyst for dehydrogenation of substituted cyclohexanones to phenols and H 2 without oxidants and hydrogen acceptors - Chemical Science (RSC Publishing) DOI:10.1039/C5SC01044F
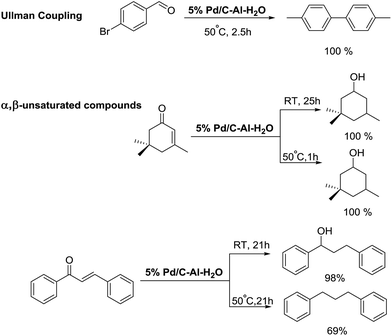
Pd/C–Al–water facilitated selective reduction of a broad variety of functional groups - Green Chemistry (RSC Publishing) DOI:10.1039/C6GC03032G
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img07.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img08.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img05.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
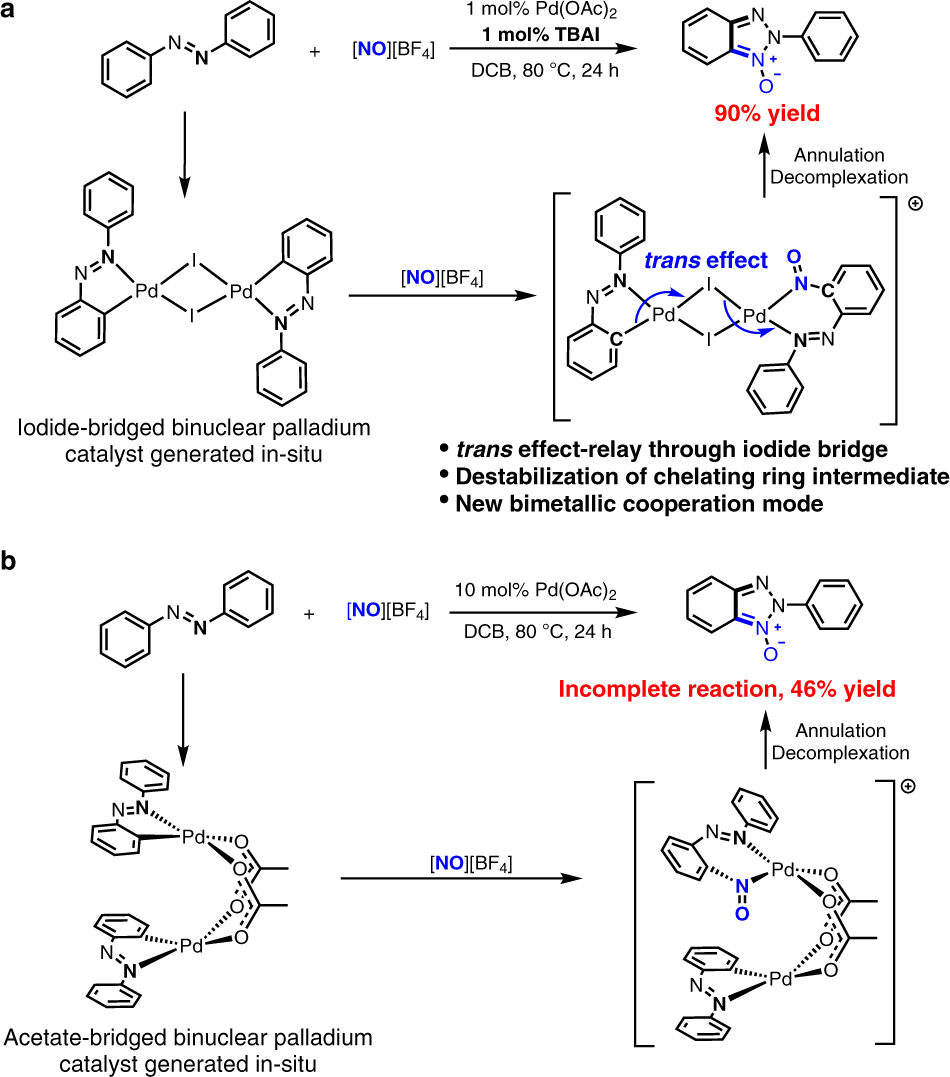
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry